Resonance and delocalisation of electrons generally leads to the stability of benzene molecule.
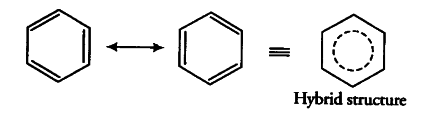
The dotted circle in the hybrid structure represents the six electrons which are delocalised between the six carbon atoms of the benzene ring. Therefore, presence of delocalised \pi-electrons in benzene makes it more stable than the hypothetical cyclohexatriene.