- What kind of colloidal solution is butter? Name a technique to separate butter from cream.
- Calculate the amount of sodium sulphate require to prepare its 20% (by mass) solution in 100 g of water.
- Butter belongs to a class of gel of colloidal solution in which dispersion medium is solid and dispersed phase in liquid. Butter can be separated from cream through the process of centrifugation.
- Mass % of sodium sulphate (Na2SO4) in solution = 20%
Mass of the solvent = 100 g
Let the mass of solute (Na2SO4) = x g
Mathematically, we have
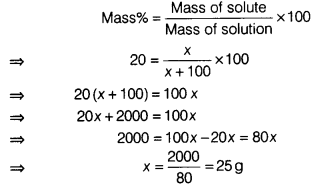
Hence, 25 g of sodium sulphate is required.