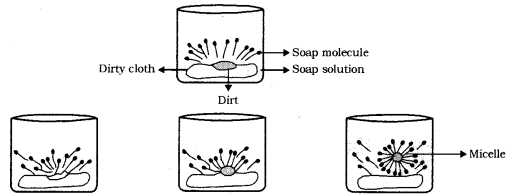
What is the difference between the molecules of soaps and detergents, chemically? Explain the cleansing action of soaps.
Soap molecules are sodium or potassium salts of long-chain carboxylic acids. They form scum upon reaction with calcium and magnesium ions present in hard water.
Detergent molecules are ammonium or sulphonate salts of long-chain carboxylic acids. They do not form insoluble precipitates with calcium and magnesium ions.
Cleansing action of Soaps: The dirt present on clothes is organic in nature and is insoluble in water. Therefore, it cannot be removed by only washing with water. When saop is dissolved in water, its hydrophobic ends attach themselves to the dirt and remove it from the cloth. Then, the molecules of soap arrange themselves in micelle formation and trap the dirt at the centre of the cluster. These micelles remain suspended in the water and, thus, the dust particles are easily rinsed away by water.