What is the difference between Sandmeyer reaction and Gatterman reaction? Explain with examples.
-
Sandmeyer Reaction
The Cl , Br and CN nucleophiles can easily be introduced in the benzene ring of benzene diazonium salt in the presence of Cu(I) ion. This reaction is called Sandmeyer reaction.
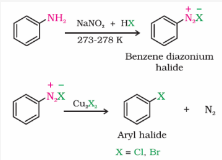
-
Gatterman Reaction
Chlorine or bromine can be introduced in the benzene ring by treating the benzene diazonium salt solution with corresponding halogen acid in the presence of copper powder. This is referred as Gatterman reaction.
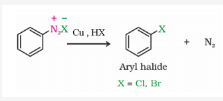
-
The yield in Sandmeyer reaction is found to be better than Gattermann reaction.
-
Gattermann reaction is a variation of Sandmeyer reaction which the CuCl is replaced by Cu powder. This replacement leads to formation of aryl halide more easily and under mild conditions than Sandmeyer reaction although the yield is reduced.