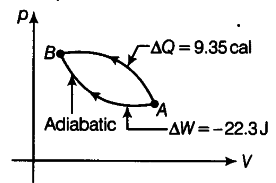
What is a cyclic process? What is change in internal energy of the system in a cyclic process? In changing the state of a gas adiabatically from an equilibrium states A to B, an amount of 22.3 J of work is done on the system. If the gas is taken from states AtoB via a process in which net heat absorbed by the system is 9.35 cat. How much is the net work done by the system in the later case? (1 cal = 4.19 J)
A cyclic process restores the system back to its initial state after completion of the cycle.
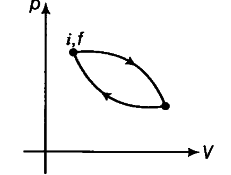
As internal energy is a state function so its value at initial point is same as that at final point (initial state is same as that of final state).