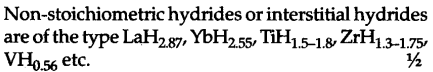
They are defident in hydrogen. These are formed by many d-block and /-block elements. No, these hydrides are not formed by alkali metals. Saline hydrides are stoichiometric, d-block or f-block elements have partially filled d or fsubshells due to which hydrogen atoms occupy interstices in the metal lattices.