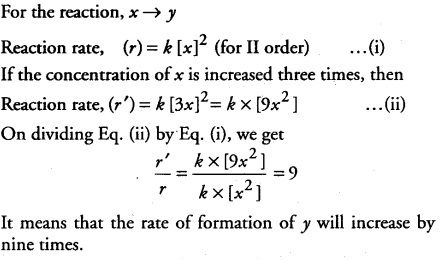The conversion of molecules x to y follows second order kinetics. If concentration of x is increased to three times, how will it affect the rate of formation of y ?
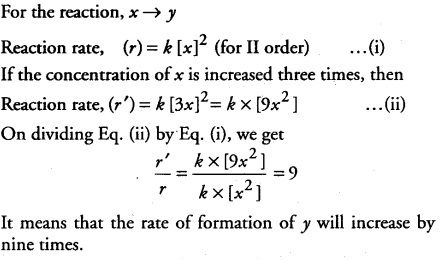
The conversion of molecules x to y follows second order kinetics. If concentration of x is increased to three times, how will it affect the rate of formation of y ?