Select the correct IUPAC name for the following branched chain alkane.

Concepts and reason
IUPAC stands for international union of pure and applied chemistry. IUPAC has given a nomenclature to name the organic compounds. The IUPAC name consists of three parts: root name, prefix and suffix.
Fundamentals
The rules for naming the compound are as follows:
- Find and name the longest continuous carbon chain.
- Identify and name groups attached to this chain.
- Number the chain consecutively, starting at the end nearest a substituent group.
- Designate the location of each substituent group by an appropriate number and name.
- Assemble the name, listing groups in alphabetical order using the full name.
(e.g. cyclopropyl before isobutyl). - The prefixes di, tri, tetra etc., used to designate several groups of the same kind, are not considered when alphabetizing.
Answer:
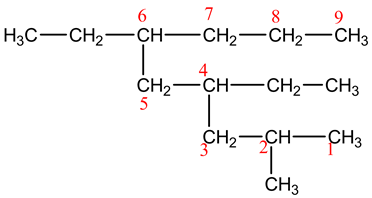
Explanation:
In this structure, there are nine carbon atoms in the long chain. Here, the functional group is alkane. So, the name of the parent chain is nonane. Number the chain and see that the substituents get the lowest possible number.
This structure contains one methyl and two ethyl substituents.

The name of the compound is 4,6-diethyl-2-methylnonane.

Explanation:
The methyl group is at 2nd carbon atom; ethyl groups are at 4th and 6th carbon atoms. So, the name of the compound is 4,6-diethyl-2-methylnonane.