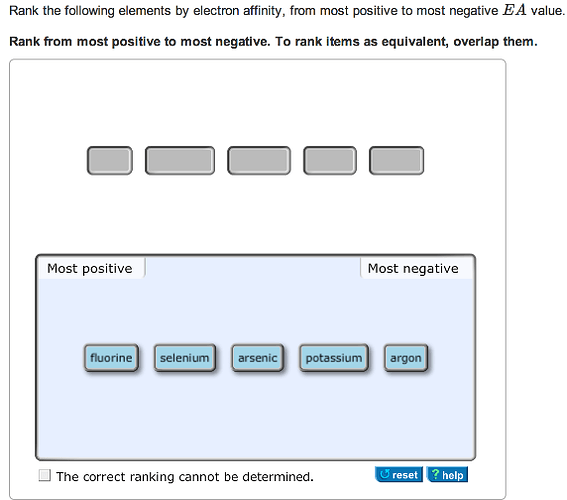
Rank the following elements by electron affinity, from most positive to most negative EA value.
Concepts and reason
The problem is based on the concept of electron affinity (EA) of an atom. It is the energy required or released to add an electron to an atom. It is the measure of the attraction between negative charge of electron and positive charge of nucleus. The factors affecting the values of electron affinities are nuclear charge, distance and screening. The stronger the attraction between the incoming electron and nucleus, the more energy is released.
Fundamentals
Electron affinities tend to become more negative moving across a period and less negative moving down a group.
There are several exceptions to the general trend in electron affinity. For example, noble gases (group 18) have most positive values and the halogens (group 17) have most negative EA values. Alkaline earth metals (group 2) have positive EA values and group 15 elements have less negative values than group 14 and group 16 elements.
Answer:
Argon is a noble gas element from group 18. Noble gases have completely filled shell, thus strongly opposes the addition of an electron. Hence, they have most positive EA values.
Potassium belongs to group 1 metals. Metals have tendency to lose electron. To add an electron they need to gain energy, hence metals are known to have lower EA values.
Arsenic belongs to group 15. It’s filled shell opposes the addition of an electron and hence has less negative EA value than its neighbor selenium from group 16.
Fluorine has the most negative EA value as it belongs to group 17 (halogens). Addition of an electron leads to noble gas configuration, which is very favorable.