In electrolysis of acidified water
why 2 vol. of hydrogen and 1 vol. of oxygen is used
In the electrolysis of acidified water,
Ionization of acidified water and electrode raections are as follows:
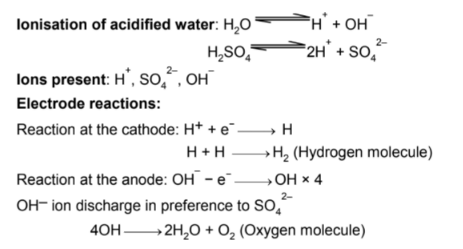
In the above reactions, it is clear that :
4 H+ ions gives 2 H2 molecules
and 4 OH- ions gives 1 O2 molecule and one water molecule.
In the electrolysis, H2: O2 ratio is 2:1