- Hydrogen is not a metal but it has been assigned a place in the reactivity series of metals. Explain.
- How would you show that silver is chemically less reactive than copper?
Answer:
- Though hydrogen is not a metal but even then it has been assigned a place in the activity series. The reason is that like metals, hydrogen also has a tendency to lose electron and forms a positive ion H+.
The metals which lose electrons less readily than hydrogen are placed below it and the metals which lose electrons more readily than hydrogen are placed above it in the reactivity series of metals. - By displacement reaction silver can be shown to be chemically less reactive than copper or copper is more reactive than silver. If a piece of silver is immersed in a solution of copper sulphate, no reaction will take place because silver is less reactive than copper and will not displace copper from the copper sulphate solution.
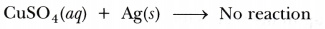
On the other hand, if a copper plate is placed in a solution of silver nitrate, copper will slowly displace silver from the solution and blue solution of copper nitrate is formed.
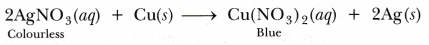
This shows that copper is more reactive than silver.