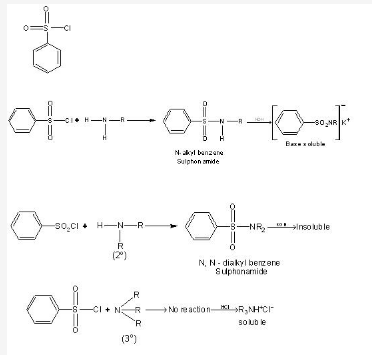
Hinsberg test is used for distinguishing primary, secondary and tertiary amines. It is used for separating 2 different amines as well.Can you please give an example for this?
The Hinsberg test, which can distinguish primary, secondary, and tertiary amines, is based upon sulfonamide formation. In the Hinsberg test, an amine is reacted with benzene sulfonyl chloride. If a product forms, the amine is either a primary or secondary amine, because tertiary amines do not form stable sulfonamides. If the sulfonamide that forms dissolves in aqueous sodium hydroxide solution, it is a primary amine. If the sulfonamide is insoluble in aqueous sodium hydroxide, it is a secondary amine. The sulfonamide of a primary amine is soluble in an aqueous base because it still possesses an acidic hydrogen on the nitrogen, which can be lost to form a sodium salt.