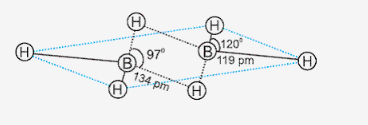
Explain structures of diborane.
- In the structure of diborane, the four terminal hydrogen atoms and the two boron atoms lie in one plane.
- Above and below this plane, there are two bridging hydrogen atoms.
- The four terminal B-H bonds are regular two centre-two electron bonds while the two bridge (B-H-B) bonds are different and can be described as three centre -two electron bonds or banana bond.