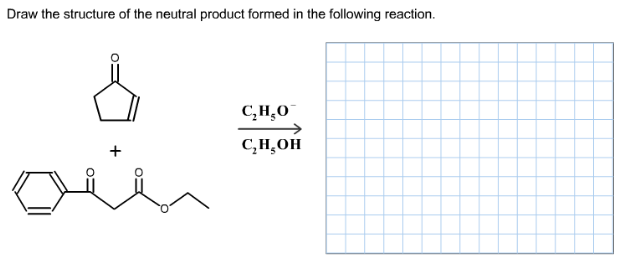
Draw the structure of the neutral product formed in the following reaction.
Concepts and reason
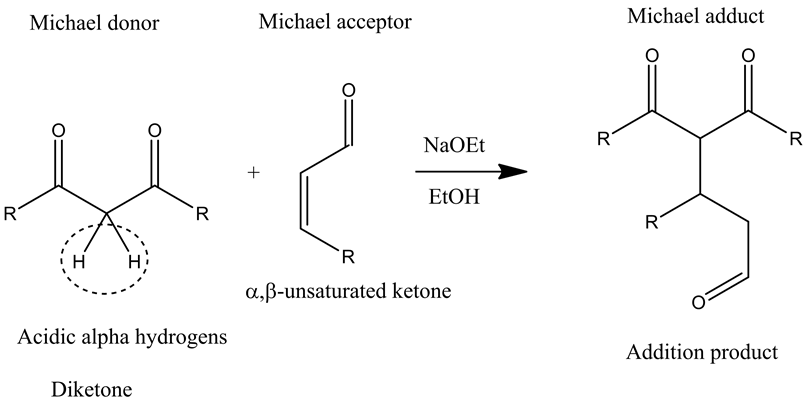
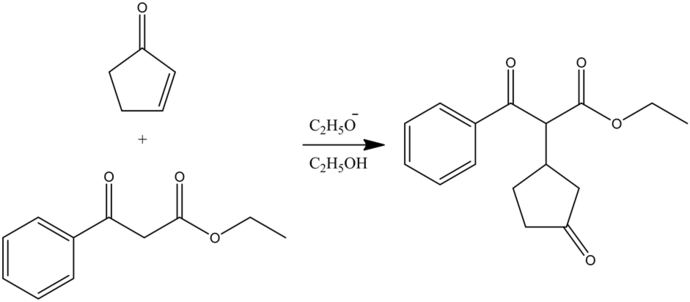
Michael Reaction
The Michael reaction or Michael addition is the organic reaction in which a carbanion as in the form of an enolate ion adds to an ![]() via conjugated addition, resulting in the formation of new carbon-carbon single bond between the Michael donor and the Michael acceptor, giving the Michael adduct as the final addition product.
via conjugated addition, resulting in the formation of new carbon-carbon single bond between the Michael donor and the Michael acceptor, giving the Michael adduct as the final addition product.
Fundamentals
Michael reaction:
Answer:
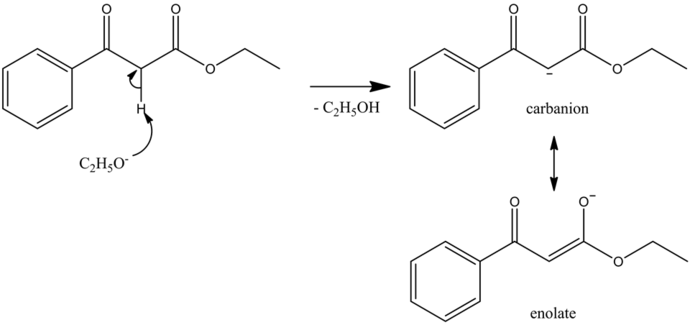
Deprotonation Step
It is an acid-base reaction. An ethoxide ion is a strong base and deprotonate the acidic alpha hydrogen present in the diketone (Michael donor) forming the carbanion which is stabilized by the resonance, giving the most stable enolate ion because the oxygen atom is more electronegative than the carbon atom.
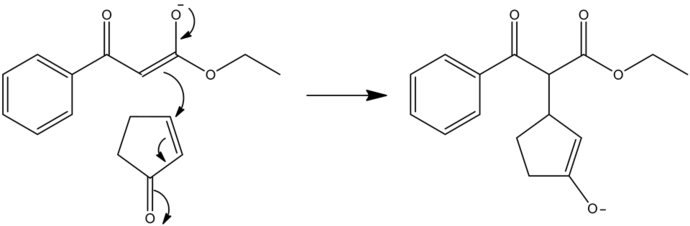
1,4-Addition Step
The nucleophilic carbanion (enolate) attacks the carbon-carbon double bond present in the ![]() via 1,4-conjugated addition, forming an intermediate enolate ion.
via 1,4-conjugated addition, forming an intermediate enolate ion.
Answer :-
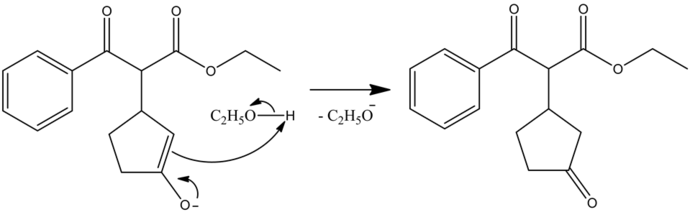
Protonation Step
Again, it is an acid-base reaction. The intermediate enolate ion is a strong base that can abstract a proton from ethanol, regenerating the ethoxide base back and gives the more stable thermodynamic product as the Michael adduct.