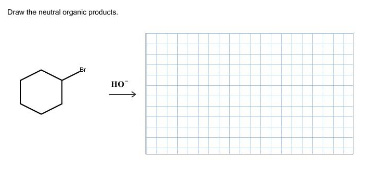
Draw the neutral organic products.
Concepts and reason
The concept used to solve this problem is ![]() . The substrate is a secondary alkyl halide and the nucleophile is strong. Elimination product is also possible to form in this reaction.
. The substrate is a secondary alkyl halide and the nucleophile is strong. Elimination product is also possible to form in this reaction.
In ,![]() the attack of nucleophile takes place from the back side, so inversion of configuration takes place in the product.
the attack of nucleophile takes place from the back side, so inversion of configuration takes place in the product.
Fundamentals
![]() reaction is a single step reaction. The rate of the reaction depends on both concentration of substrate and concentration of nucleophile.
reaction is a single step reaction. The rate of the reaction depends on both concentration of substrate and concentration of nucleophile.
E2 mechanism is favored by strong, negatively charged bases, especially OH- and OR-.
The reaction occurs with primary, secondary and tertiary alkyl halides. So, the order of the reactivity would be tertiary > secondary > primary. In E2 mechanism, all the bonds are broken and formed in a single step.
Answer:
Explanation:
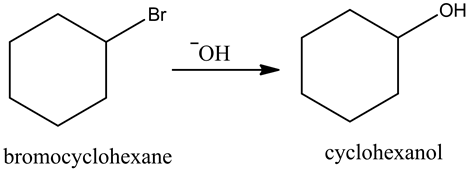
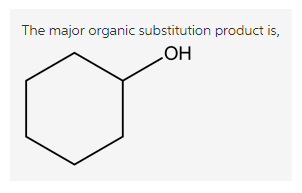
Treatment of bromocyclohexane with a strong nucleophile, hydroxide ion, gives cyclohexanol. Here, the hydroxide attacks from the back side of the carbon having leaving group, the C-Br bond cleaves and forms new C-O bond in the product.
The reaction is as follows:
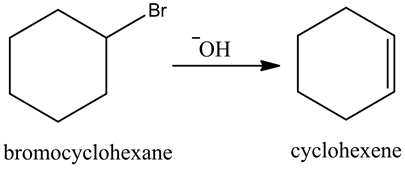
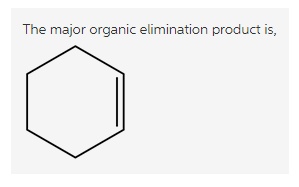
Explanation:
Treatment of bromo cyclohexane with a strong base, gives cyclohexene. Here, the base abstracts the beta hydrogen atom and forms a double bond in the product.