Draw the major organic product formed in the following reaction.
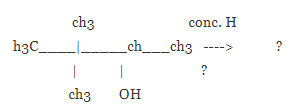
Concepts and reason
Alcohols undergo a dehydration reaction when the react with concentrated acids in the presence of heat. Product formation is undergone with the help of an elimination reaction.
Fundamentals
When alcohols undergo a reaction with strong acids (like H2SO4, H3PO4) in the presence of heat, it forms an alkene product with the elimination of water. It undergoes 1, 2 –elimination to form an alkene.
Example:
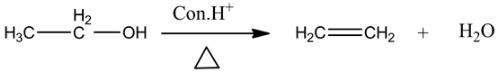
Answer:
Explanation:
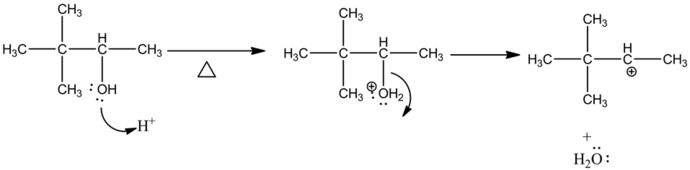
The alcoholic oxygen undergoes protonation with the help of a lone pair of electrons and makes it a better leaving group. Then the leaving group cleaves and forms a carbocation intermediate which is the rate determining step for this reaction.
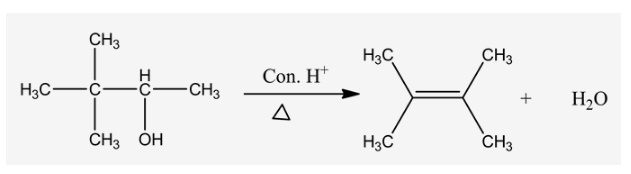
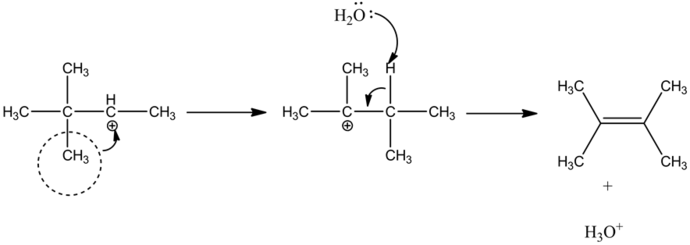
Explanation:
The methyl group is rearranged and forms carbocation in the second carbon. Then the lone pair of oxygen electrons abstracts a proton and forms an alkene product by stabilizing the carbocation.