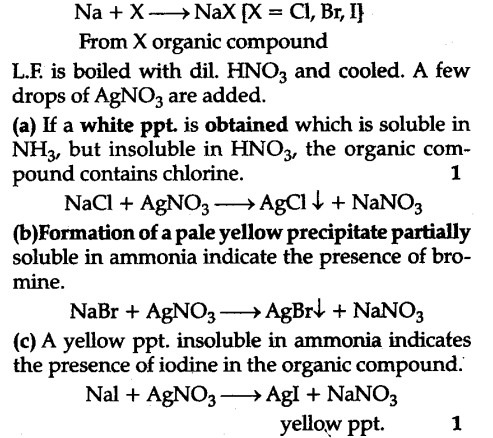Preparation of Lassaigne’s filterate. A pea-sized and dried fresh piece of sodium is put in a small ignition tube and heated dll sodium melts. A pinch of the given organic compound (or a drop of it if it is a liquid) is now added to the fused sodium and heated again to red hot. The fusion tube contents are now transferred to a china dish containing 10-15 ml of water. The tube breaks arid contentsare boiled in the China dish. The solution, after filtration, is called Lassaigne’s filterate. It is used for testing of the elements, N, S, N and S present together phosphorus and halogens in the given organic compound.
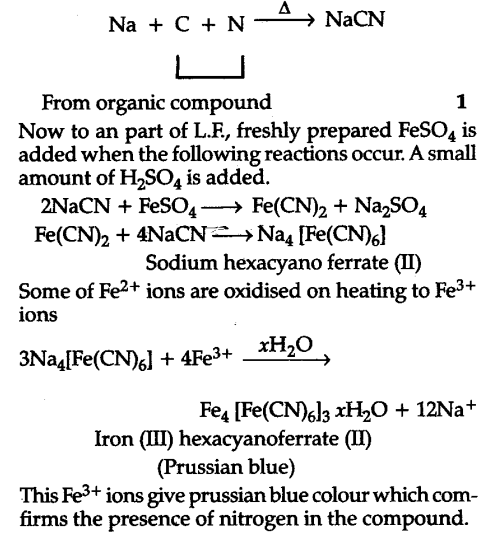
Test for Nitrogen
Test for N and S if present together
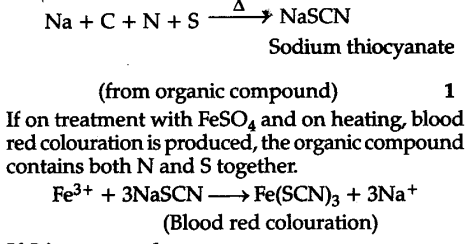
If S is present alone
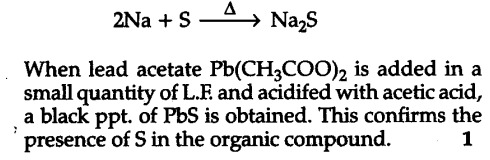
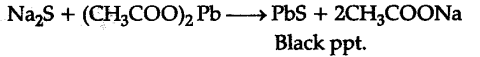
Test for Halogens