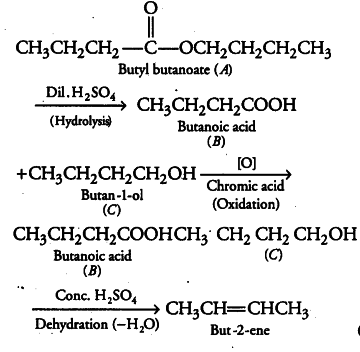
An organic compound A (molecular formula was hydrolysed with dilute sulphuric acid to give a carboxylic acid (B) and alcohol ©. Oxidation of © with chromic acid produced (B). C on dehydration gives but-2-ene. Write equations for the reactions involved.
(i) Since, A produces carboxylic acid B and an alcohol C on hydrolysis, hence compound A is an ester.
(ii) Alcohol C on oxidation produces acid B. It means both B and C have same number of carbon atoms, i.e. four each as C on dehydration gives but-2-ene.
The equations for all the above reactions are: