An element X which is a yellow solid at room temperature shows catenation and allotropy. X forms two oxides which are also formed during the thermal decomposition of ferrous sulphate crystals and are the major air pollutants.
- Identify the element X.
- Write the electronic configuration of X.
- Write the balanced chemical equation for the thermal decomposition of ferrous sulphate crystals.
- What would be the nature (acidic/basic) of oxides formed?
- Locate the position of the element in the Modern Periodic Table.
Answer:
- Element X is sulphur (atomic no. 16)
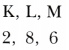
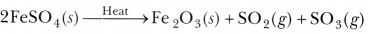
- Acidic
- 3rd period, group 16