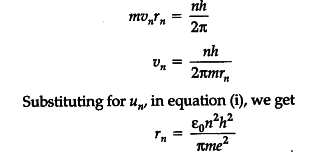(a) Write two important limitations of Rutherford model which could not explain the observed features of atomic spectra. How were these explained in Bohr’s model of hydrogen atom ? Use the Rydberg formula to calculate the wavelength of the ${ H }_{ \alpha }$ line. (Take R = 1.1 x ${ 10 }^{ 7 }{ m }^{ -1 }$ ).
(b) Using Bohr’s postulates, obtain the expression for the radius of the ${ n }^{ th }$ orbit in hydrogen atom. [Delhi I, II, III 2015]
(a) (i) Electron moving in a circular orbit around the nucleus would get accelerated, therefore it would spiral into the nucleus, as it looses its energy.
(ii) It must emit a continuous spectrum.
According to Bohr7 s model of hydrogen atom,
(i) Electron in an atom can revolve in certain stable orbits without the emission of radiant energy.
(ii) Energy is released/absorbed only, when an electron jumps from one stable orbit to another stable orbit. This results in a discrete spectrum.