A generic gas, X, is placed in a sealed glass jar and decomposes to from gaseous Y and solid Z.

Answer:
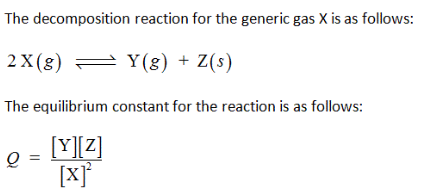
(a)
As per the le chatelier principle, increase in reactant concentration will shift equilibrium towards right side. If concentration of X changes then concentration of Y and Z also changes to reach the equilibrium.
Mass of Z is directly proportional to the concentration of Z, so mass of Z changes if initial concentration changes.
If initial concentration of x changes then concentration of X at equilibrium also changes.

(b)
The total pressure of the system at equilibrium changes with initial concentration because as the concentration changes then number of moles also changes. If number of moles of X changes then total pressure of the system changes.
