A compound which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water. Identify the compound. Write the chemical equation for its preparation. For what purpose is it used in hospitals?
Answer:
The compound prepared from gypsum on heating it at 100°C, is known as Plaster of Paris. Its chemical formula is CaS04.f H20. Hence, its chemical name is calcium sulphate hemihydrate. The chemical equation for its preparation is as follows:
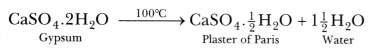
Plaster of Paris is used in hospitals mainly as plaster for supporting fractured bones in the right position. In dentistry, it is used for making casts.