Using VSEPR theory, draw the molecular structures of ${ OSF }{ 4 }$ and
${ XeF }{ 4 }$ indicating the location of lone pair (s) of electrons and hybridisation of central atoms.
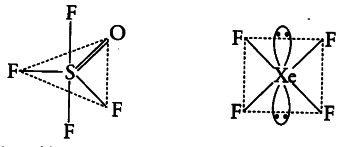
Using VSEPR theory, draw the molecular structures of ${ OSF }{ 4 }$ and
${ XeF }{ 4 }$ indicating the location of lone pair (s) of electrons and hybridisation of central atoms.
