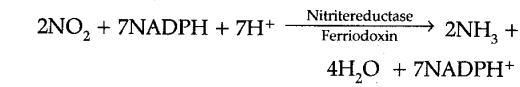Nitrate is the most important source of nitrogen for the plants. However, it cannot be used as such by the plants in the synthesis of organic compounds.
It is first reduced to ammonia before being incorporated into organic compounds. The process of nitrate reduction to ammonia is carried out in two steps, each mediated by a specific enzyme.
(i) Reduction of Nitrate to Nitrite : (a) The nitrate is reduced to nitrite by an enzyme called nitrate j reductase, (b) The enzyme contains FAD as ! prosthetic group which receives hydrogen from reduced coenzyme (NADH/NADPH + H+) for the reduction of the nitrate, © The reduced coenzyme (NADH / NADPH+ H+) serves as hydrogen donor and molybdenum serves as an electron carrier in j the process.
(ii) Reduction of Nitrite : (a) The nitrite ions are then reduced to ammonia by an enzyme called nitrite reductase, (b) This enzyme requires copper and iron for its activity, © The enzyme nitrate reductase occurs inside chloroplasts in the leaf cells and leucoplasts of other cells. The reduced coenzyme (NADPH + H+) serves as hydrogen donor in illuminated cell and NAD + H+ in others for the reduction of nitrites, (d) The process of reduction also requires ferriodoxin which occurs in the higher plants mostly in the leaves.