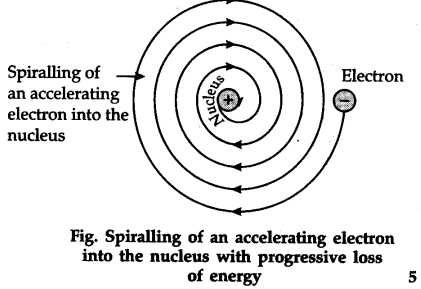
(i) Drawbacks of Rutherford’s atomic model— According to Rutherford’s model, electrons are orbiting the nucleus ; hence, the direction of their velocity is constantly changing, i.e., the electrons are accelerating. This will cause the electrons to emit or radiate energy; and consequently, the electrons will have lesser and lesser energy will get closer and closer to the nucleus, until at last it spirals into the nucleus.
(ii) An electron moving inward along a spiral path must continuously radiate energy. However in actual practice spectral lines of fixed wavelength are observed.