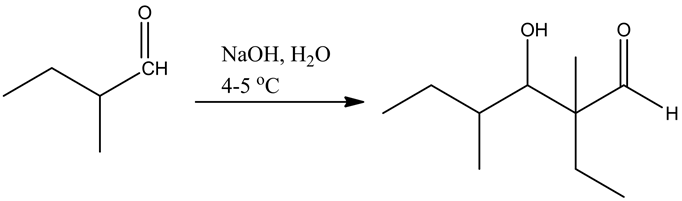
Draw the structure of the product formed when 2-methylbutanal is treated with cold aqueous base.
Concepts and reason
- A condensation reaction is a type of chemical reaction, which has a huge importance in an organic synthesis.
- The reaction undergoes the formation of a carbon-carbon double bond. An aldol condensation reaction is a type of condensation reaction, in which two aldehydes have α-hydrogen atom, which react with each other to form an aldol product.
- The first step is the aldol reaction, in which the enolate ion is formed, then it reacts with another molecule of the aldehyde.
- The dehydration of product of an aldol reaction, forms the αβ-unsaturated carbonyl molecules (aldol). This is known as an aldol condensation reaction.
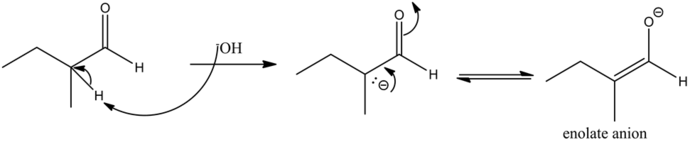
- In a base catalyzed aldol reaction, the base deprotonates the acidic hydrogen on an α-carbon atom to form the enolate.
- An enolate is a good nucleophile and carbonyl is a good electrophile. So, the enolate reacts with the carbonyl carbon of another aldehyde.
Fundamentals
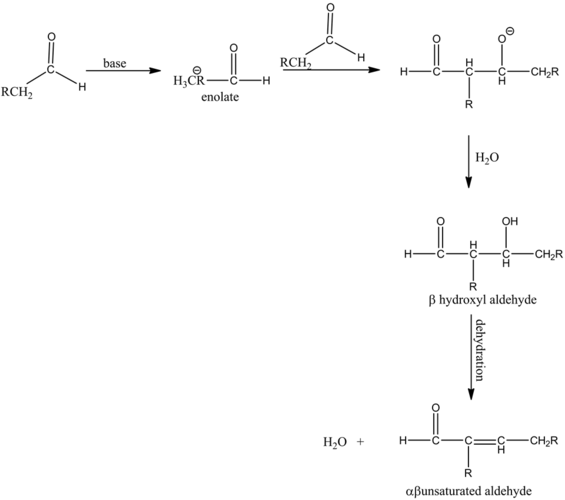
The mechanism of an aldol condensation contains of two steps. The first part is an aldol reaction and the second part is a dehydration elimination reaction. First, the base abstracts the proton to form a carbanion, and then it attacks the carbonyl group of another compound to form an oxy anion. This compound undergoes a hydrolysis and forms an aldol, which is followed by dehydration that will give an aldol product.
Mechanism:-
Answer:
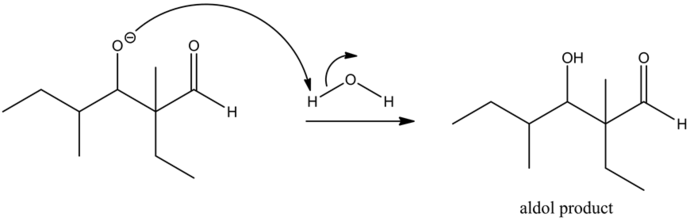
The base abstracts the alpha hydrogen of aldehyde to form a carbanion. The carbanion donates its electrons to form a highly resonance-stabilized enolate anion. The enolates are very strong nucleophiles.
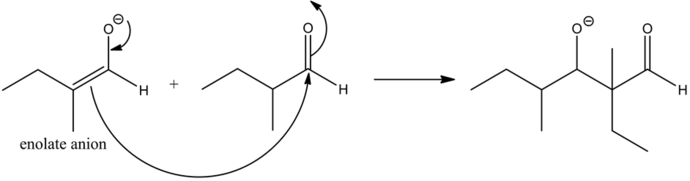
The enolates are very strong nucleophiles and the carbon of carbonyls is a good electrophile by nature. Hence, the nucleophilic carbon from the enolate, attacks the electrophilic carbonyl carbon, giving it a condensed product.
The structure of the product that is formed, when 2-methylbutsnal is treated with a cold aqueous base is given below:
The reaction of the alkoxide with water molecule gives the aldol condensation product between two aldehydes. The dehydration of the aldol product does not occur to form enol, since there is no alpha hydrogen that is present with respect to the carbonyl group.