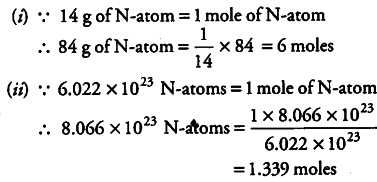Calculate the number of moles for the following:
(i) 84 g of nitrogen atoms
(ii) 8.066 x ${10}^{23}$ number of N-atoms
[Atomic mass of N = 14]
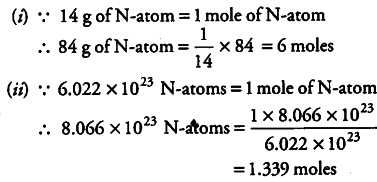
Calculate the number of moles for the following:
(i) 84 g of nitrogen atoms
(ii) 8.066 x ${10}^{23}$ number of N-atoms
[Atomic mass of N = 14]