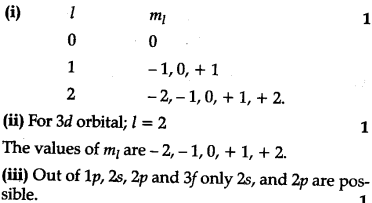(i) An atomic orbital has n = 3. What are possible value of l and ${{m}{l}}$?
(ii) List the quantum numbers ${{m}{l}}$ and /) of electrons for 3d orbital.
(iii) Which of the following orbitals are possible? Ip, 2s, 2p and 3 f.
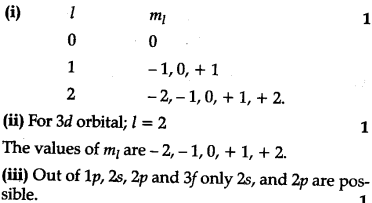
(i) An atomic orbital has n = 3. What are possible value of l and ${{m}{l}}$?
(ii) List the quantum numbers ${{m}{l}}$ and /) of electrons for 3d orbital.
(iii) Which of the following orbitals are possible? Ip, 2s, 2p and 3 f.