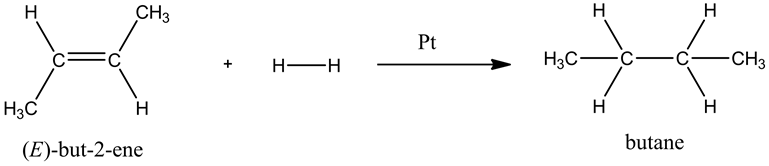
Draw the product of the following reaction. Show all the hydrogen atoms in your structure.
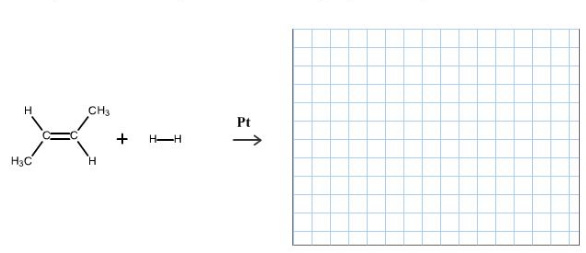
Concepts and reason
Alkenes react with dihydrogen in presence catalysts like platinum, nickel, and palladium carbon to form an alkane. This process is called catalytic hydrogenation.
Fundamentals
Catalytic hydrogenation is surface phenomenon. In this reaction, both hydrogens add to the double bond from the same side.
Answer:
The reactants are (E)-but-2-ene and dihydrogen.
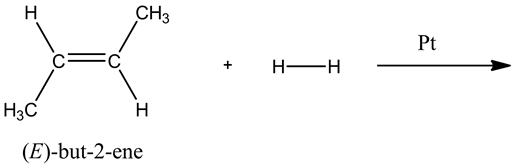
Explanation:
The reactants are (E)-but-2-ene and dihydrogen ![]() , and platinum acts as catalyst.
, and platinum acts as catalyst.
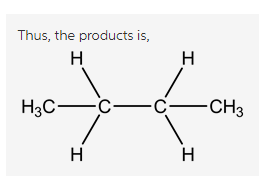
Explanation:
(E)-but-2-ene undergoes reduction in presence ![]() to form butane as a product.
to form butane as a product.