
Write two uses of plaster of paris.

Write two uses of plaster of paris.
(a) The alkali metal ions are highly hydrated. The smaller the size of the ion, the greater is the degree of hydration. The Li+ ion gets most hydrated and Cs+ ion least.
The result in decrease in hydrated ionic radii in the order :
Li+ > Na+ > K+ > Rb+ > Cs+
As a result, the hydrated Li+ ion being largest in ionic size has the lowest mobility in water and Cs+ ion has the highest mobility and follow the order
Li+ < Na+ < K+ < Rb+ < Cs+
(b)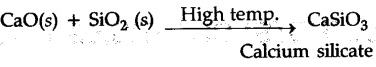
©© Uses of plaster of paris :
(i) It is used for casting statues and busts.
(ii) It is used for immobilising the affected part of the organ where there is bone fracture or sprain.