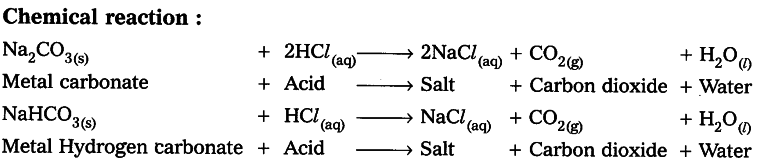
Write an activity to show that all metal carbonates and hydrogen carbonates react with acids to give a corresponding salt.
Aim : To show that all metal carbonates and hydrogen carbonates react with acids to give a corresponding salt.
Required Materials :
- Two test tubes
- Sodium carbonate
- Sodium hydrogen carbonate
- Two holed rubber stopper
- Thistle funnel
- Stand
- Dilute hydrochloric acid
- Delivery tube
- Calcium carbonate (in a test tube)
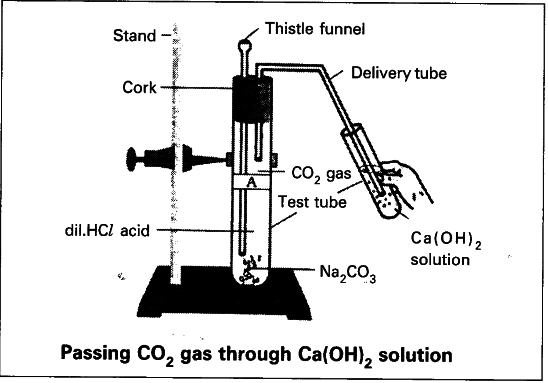
Procedure : - Take a test tube A with 0.5 gm of sodium carbonate.
- Close the test tube A with two holed rubber stopper.
- Insert a thistle funnel through one hole and insert a delivery tube through the other hole.
- Pour 2ml of dilute HCl to the test tube A.
- Do the same as above with test tube B with sodium hydrogen carbonate. Observation : Carbon dioxide is released from test tube A and B.
Result: All metal carbonates and hydrogen carbonates react with acids to give a corresponding salt.