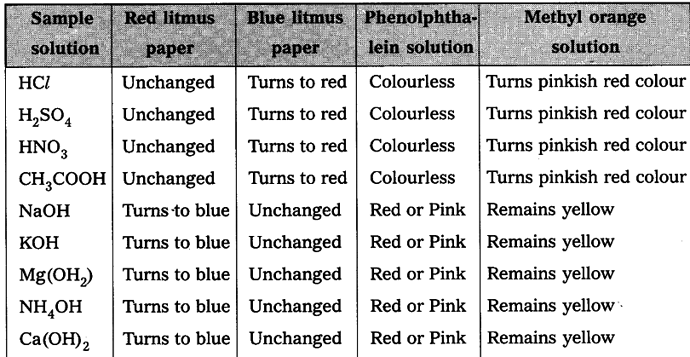
Observe the change in colour in each case and tabulate the results in the table
Producure :
- Collect the following samples from the science laboratory.
- Hydrochloric acid (HCl)
- Sulphuric acid (H_{2}SO_{4}
- Nitric acid (HN{ O }_{ 3 })
- Acetic acid (CH_{3}COOH)
- Sodium hydroxide (NaOH)
- Calcium hydroxide [Ca(OH)2]
- Magnesium hydroxide [Mg(OH)2]
- Ammonium hydroxide (N{ H }_{ 4 }OH)
- Potassium hydroxide (KOH)
- Prepare dilute solutions of the respective substances.
- Take four watch glasses.
- Put one drop of the first solution in each one of them and test the solution as follows.
- Dip the blue litmus paper in the first watch glass.
- Dip the red. litmus paper in the second watch glass.
- Add a drop of methyl orange to the third watch glass.
- Add a drop of phenolphthalein to the fourth watch glass.
Observation : Observe the respective colour changes and note down in the chart below.