In the study of Geiger-Marsdon experiment on scattering of a-particles by a thin foil of gold, draw the trajectory of a-particles in the Coulomb field of target nucleus. Explain briefly how one gets the information on the size of the nucleus from this study. From the relation and A is the mass number of the nucleus, show that nuclear matter density is independent of A.
Trajectory of a-particles in the coulomb field of target nucleus is shown below:
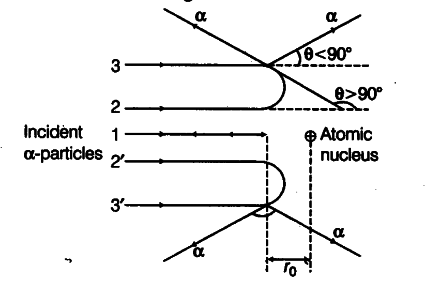
From this experiment, the following points are observed.
(i) Most of the particles pass straight through the gold foil. It means that they do not suffer any collision with gold atoms.
(ii) About one particle in every 8000 particles
deflects by more than 90°. As most of the particles go undeflected and only a few get deflected, this shows that most of the space in an atom is empty and at the centre of the atom, there exists a nucleus. By the number of particles get deflected, the information regarding size of the nucleus can be known.