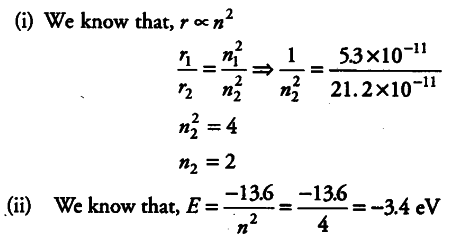In the ground state of H-atom, its Bohr’s radius is given as 5.3 x ${{10}^{-11}}$ m . The atom is excited such that the radius becomes 21.2 x 10“u m . Find (i) the value of the principal quantum number and (ij) the total energy of the atom in this excited state.