How do you account for the strong reducing power of lithium in aqueous solution?
Electrode potential is a measure of the tendency of an element to lose electrons in the aqueous solution. It mainly depends upon the following three factors, i.e.,
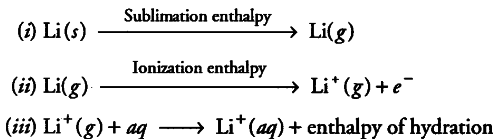
With the small size of its ion, lithium has the highest hydration enthalpy. However, ionization enthalpy of Lr is highest among alkali metals but hydration enthalpy predominates over ionization enthalpy. Therefore, lithium is the strongest reducing agent in aqueous solution mainly because of its high enthalpy of hydration.