${{H}{3}}P{{O}{3}} can be represented by structures 1 and 2 shown below. Can these two structures taken ' as the canonical forms of the resonance hybrid representing {{H}{3}}P{{O}{3}}$ ? If not, give reasons for the same.
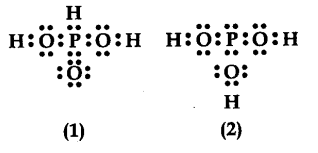
No the structures (1) and (2) cannot be taken as canonical forms of ${{H}{3}}$P${{O}{3}}$ because the positions of atoms have been changed.
Two structures to represent resonance should have same positions of atoms.