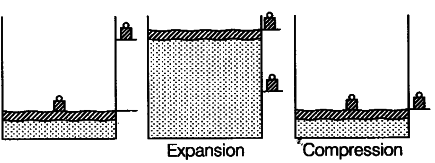
Explain with the suitable example that a reversible process must be carried slowly and a fast process is necessarily irreversible.
A reversible process must pass through equilibrium states which are very close to each other so that when process is reversed, it passes back through these equilibrium states and then it is again decompressed or it passes through same equilibrium states, system can be restored to its initial state without any change in surroundings.
e.g., If a gas is compressed as shown
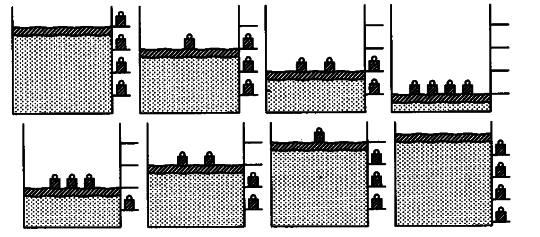
But a reversible process can proceeds without reaching equilibrium in intermediate states.