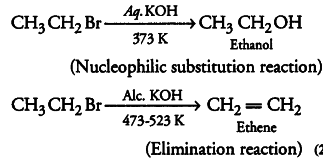
Elimination reactions (especially beta-elimination) are as common as the nucleophilic substitution reaction in case of alkyl halides. Specify the reagents used in both cases.
Elimination reactions are as common as the nucleophilic substitution reaction in case of alkyl halides as two reactions occur simultaneously. Generally, at lower temperature and by using weaker base, nucleophilic substitution reactions occur while at higher temperature and by using a stronger base, elimination reactions (especially beta-elimination) take place.
e.g. If ethyl bromide is treated with aq. KOH at low temperature, it gives ethanol while if it is treated with ale. KOH at high temperature then it gives ethene.