(i) Describe the following for getting alkanes: (a) Wurtz reaction
(b) Decarboxylation of Na-salts of fatty acid
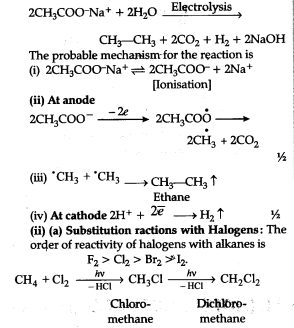
© Kolbe’s electrolytical method,
(ii) Also write with suitable examples the following reactions of alkanes:
(a) Substitution reactions
(b) Oxidation by combustion
© Isomerisation
Methods of preparation of Alkanes :
(i) (a) Wurtz reaction: Alkyl halides on treatment with sodium in dry ether gives higher alkanes, preferably containing even number of carbon atoms.
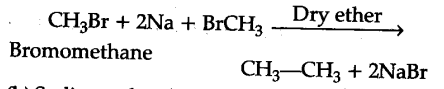
Sodium salts of fatty acids on heating with soda
lime (a mixture of NaOH and CaO) give alkanes
containing one carbon atom less than the carboxylic acid. The process of elimination of carbon dioxide from a carboxylic add is known as Decarboxylation
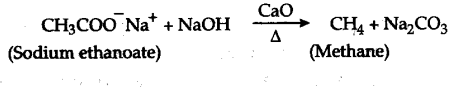
Kolbe’s electrolytical method : An aqueous solution of sodium or potassium salt of a carboxylic add on electrolysis gives alkanes containing even number of carbon atoms at anode.