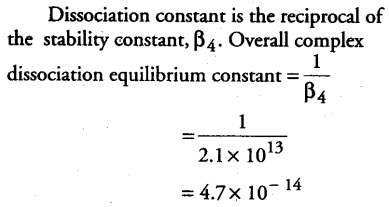Calculate the overall complex dissociation equilibrium constant for the [Cu(NH 3)4]2+ ions, given that beta 4 for this complex is 2.1 x ${{10}^{13}}$
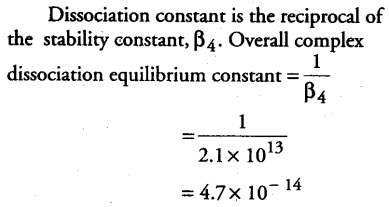
Calculate the overall complex dissociation equilibrium constant for the [Cu(NH 3)4]2+ ions, given that beta 4 for this complex is 2.1 x ${{10}^{13}}$