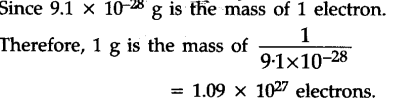(i) Calculate the number of electrons which will together weigh one gram.
(ii) Calculate the mass and^charge of one mole of electrons.
[1 mole species = 6.022 x ${{10}^{23}}$ atom Mass of 1${{e}^{-}}$ = 9.11 X ${{10}^{-31}}$ kg
Charge on 1${{e}^{-}}$ = 1.602 x ${{10}^{-19}}$ C]