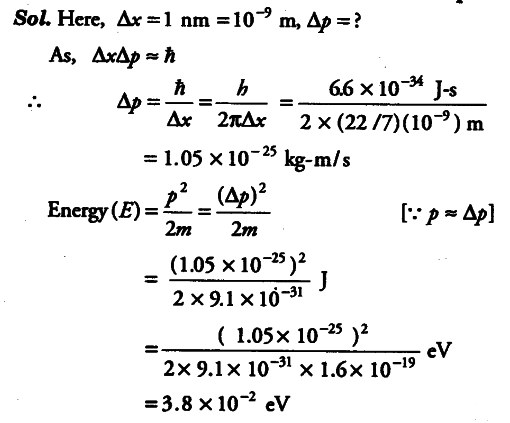Assuming an electron is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg uncertainty principle .You can assume the uncertainty in position Ax as 1 nm. Assuming p = ∆p, find the energy of the electron in eV.
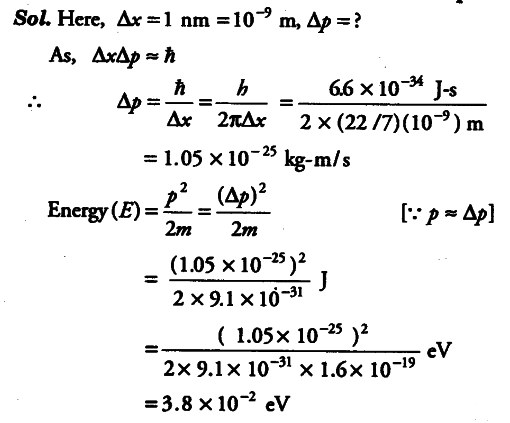
Assuming an electron is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg uncertainty principle .You can assume the uncertainty in position Ax as 1 nm. Assuming p = ∆p, find the energy of the electron in eV.