(a) Liquid drops are spherical in shape. Why ?
(b) Derive the expression of excess pressure inside the liquid drop.
© Give two similarities and dissimilarities between
friction and viscosity.
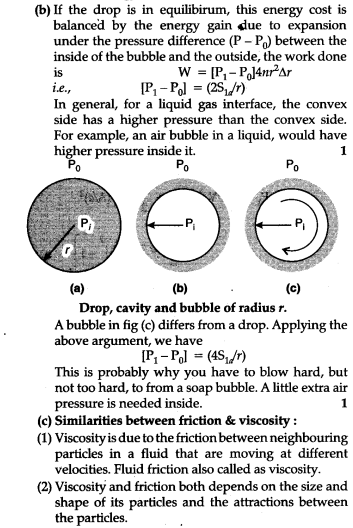
(a) Liquid drops : One consequence of surface tension is that free liquid drops and bubbles are spherical if effects of gravity can be neglected. You must have seen this clearly in small drops just formed in a high speed spray or in a shop bubbles blow, by most of us in the childhood. Why are drops and bubbles spherial ? What keeps soap bubbles stable ?
As we have been saying repeatedly, a liquid air interface has energy, so for a given value the surface with minimum energy is the one with the least area. The sphere has this property. Though it is out of the scope of this book, but you can check that a sphere is better than a cube in this respect. So, if gravity and outer forces were ineffective, liquid drops would be spherical. Another interesting consequence of surface tension is that the pressure inside a spherical drop is more that the pressure outside. Suppose a spherical drop of radius is in equilibrium. If its radius increase by the extra surface energy.

these images from which book, if possible to tell me book name ?