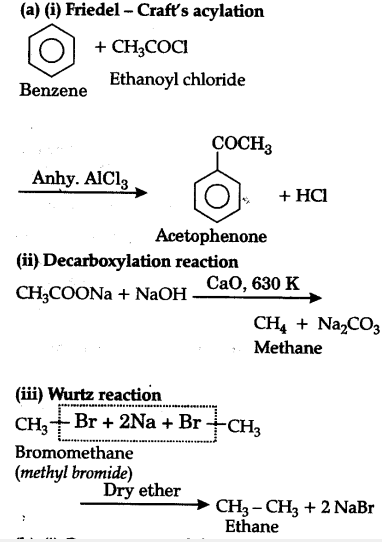
(a) Give the chemical equation of the following reactions:
(i) Friedal-Craft’s acylation,
(ii) Decarboxylation reaction,
(iii) Wurtz reaction.
(b) Account for the following:
(i) Benzene is extra-ordinarily stable though it contains three double bonds.
(ii) Branching of alkane chain decrases boiling point.
(b)(i) Resonance or delocalization of electrons usually leads to stability. Since, in benzene all the six electrons of the three double bonds are completely delocalized to form one lowest energy molecular $\pi $orbital which surrounds all the carbon atoms of the ring, therefore, it is extraordinarily stable.
(ii) Amongst isomeric alkanes, the branched chain isomer has invariably the lower boiling point than the corresponding n- alkane. This is due to the reason that with braching the shape of the molecule tends to approach that of a sphere. As a result, the surface area of the branched isomer decreases. Due to the lesser surface area of these molecules, the van der Waals forces of attraction operating between their molecules become comparativelyweaker and hence lesser amount of energy is re-quired to overcome (hem. Asaresult, the bdBngpoints erf branched chain isomers are lower than those of the corresponding n-alkanes. e.g.